|
Updates Contents |
|
Plenty of updates.
Section 5.3 (Body-Build)
A. G. Alias reported elevated body hair but a weaker muscular build in a sample of homosexual men compared to general population-matched male controls.(1) See Fig 1 below. Other studies cited in the book mention the weaker muscular build of homosexual men, and this is often observed anyway in large gatherings of homosexual men.
Alias mentioned reports of elevated body hair among male homosexuals by both Magnus Hirschfeld(2) and Henry.(3) Hirschfeld reported unusual body hairiness in 30% of 500 German homosexual men,(2) although a comparable level of body hairiness existed only among 10% of the general white male population.(4) Hirschfeld’s observations carry a lot of weight because he observed thousands of homosexual males. In both the samples of Hirschfeld and Henry, as well as that of Alias, about 20% of the male homosexuals had very little body hair. Therefore, given the rarity of homosexual men, differences with respect to body hairiness between heterosexual and homosexual men will not be readily observable, and have come to light only after systematic observations.
The participants were a non-random sample of 420 men and 342 women, aged 36-40 years, recruited to answer a sexuality/sexual-orientation-across-the-lifespan survey via advertisements in print publications, announcement to community groups, and the internet. Of the 513 packets sent to 15 community groups for distribution, 26.7% were returned, and of the 73 phone requests for questionnaires to be sent by mail, 32.9% were returned.
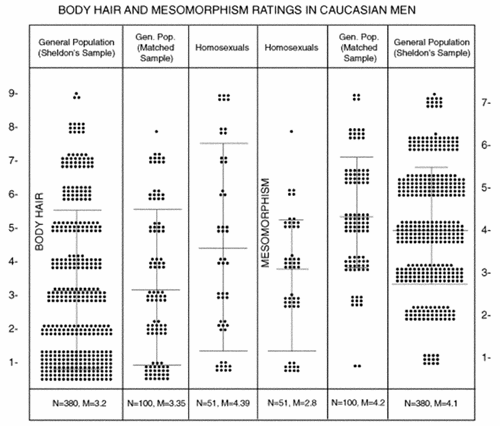
Fig 1: “Fifty-one Caucasian homosexual men’s half-naked (Polaroid) pictures were compared with 100 fairly matched, similar pictures from the general population and with all 380 clearly printed Caucasian men’s pictures from Sheldon’s9 “Atlas of Men,” aged 25–54. Body hair ratings were done by two judges jointly, using three reference sketches, from 1–9. Mesomorphism (Mes) ratings were done by assessing muscular development or the potential for it, except in Sheldon’s sample where Sheldon's own Mes numbers were used. Seven typical Sheldon’s pictures, representative of each Mes number, from 1–7, were used. Each Polaroid picture in both groups was rated jointly by the same two judges, from 1–7, by careful comparison with each of the seven Sheldon pictures. The homosexual men were more hirsute (P <0.01) but less muscular (P <0.001) than both controls. The controls were not different in either body hair or Mes ratings.”(1)
Fig 2 shows extreme contrasts between homosexual men (top row) and heterosexual men (bottom row) in the samples of Alias.
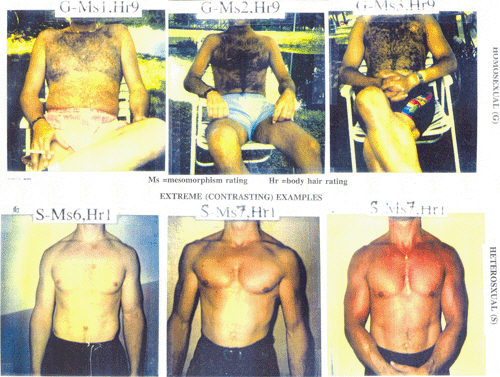
Fig 2: Extreme contrasts between homosexual and heterosexual men with respect to body hair and muscularity in the samples of Alias (pictures courtesy of A. G. Alias).
Both Hirschfeld(2) and Henry(3) have also noted the more manly build of lesbians compared to heterosexual women.
Elevated body hair among homosexual men is consistent with a larger penis in male homosexuals compared to heterosexual men. Since body hair and the penis are strongly dependent on dihydrotestosterone (DHT) for growth, and DHT formation is a way to amplify the effect of testosterone on select target tissues, the report by Alias mentions yet another hypermasculine correlate of male homosexuality. Since DHT is also implicated in male-pattern baldness and acne, a comparison between homosexual men and heterosexual men with respect to these variables is relevant.
As addressed in the book, androgens can be expected to influence various simple structures in a dose-response manner, but beyond a level that a developing organism can handle, the developmental disturbances caused by excess androgens will not result in a dose-response effect but a non-linear effect on complex structures (which require a longer time and a more stable developing environment to develop properly). Thus, excess androgens from fetal development onward could result in hypermasculine forms of simple structures such as body hair or penis but hypomasculine forms of more complex structures such as the musculoskeletal system and certain brain structures. In this regard, consider Fig 3 below.
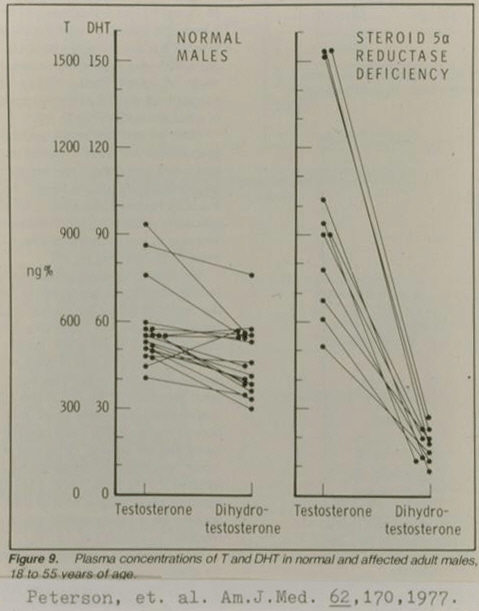
Fig 3: Testosterone and dihydrotestosterone (DHT) in normal men and men with 5-alpha reductase-2 deficiency. Note that men with 5-alpha reductase-2 deficiency still produce some DHT as a result of 5-alpha reductase-1 (scan courtesy of A. G. Alias).
As can be seen from Fig 3 above, only a small fraction of testosterone is converted to DHT. Therefore, if the find in Fig 1 above is to be understood in terms of greater conversion of testosterone to DHT among homosexual men, testosterone being relevant to muscularity, homosexual men would still have plenty of testosterone left to manifest roughly similar muscularity to that of heterosexual men. Along these lines, after being confronted by a strange mix of feminine, hypomasculine, normomasculine, and hypermasculine traits among homosexual men, Rahman and Wilson have proposed that since testosterone-derived-estradiol is implicated in mammalian defeminization, male homosexuality is caused by reduced aromatization of testosterone to estradiol, the extra testosterone being responsible for the hypermasculine correlates of homosexuality.(5) However, only a miniscule fraction of testosterone in males is converted to estradiol (see note below), i.e., reduced aromatization of testosterone is unlikely to substantially increase testosterone levels (besides, as discussed in the book, estradiol appears to exert a weaker defeminizing influence on humans compared to rodents), and as has been reported repeatedly, homosexual men manifest a less muscular build than heterosexual men, on average. Therefore, after an evaluation of numerous feminine, hypomasculine, normomasculine, and hypermasculine correlates of male homosexuality--as addressed in the book--in addition to this new hypermasculine correlate of male homosexuality reported by Alias, it appears that male homosexuality is related to excess prenatal testosterone exposure, where excess is in the sense of beyond one’s handling capability, which in turn is consistent with considerable evidence listed in the book that male homosexuality is related to prenatal developmental disturbances.
Note: The testiticular testosterone secretion rate is 4-9 mg/d (13.9-31.33 umol/d) in normal adult men, among whom total plasma testosterone level is 300-1000 ng/dL (10.4-34.7 nmol/L), of which 2% is free testosterone. In normal adult men, the total estradiol production rate is approximately 50 ug/d (184 nmol/d) and the plasma estradiol level is 20-50 pg/mL (73-184 pmol/L), of which 2% is free. The values during fetal development will obviously be different but the substantial difference in the amount of testosterone and estradiol production is likely to be of a similar order of magnitude.
References:
- A. G. Alias, Ann N Y Acad Sci 1032, 237 (Dec, 2004).
- M. Hirschfeld, in Die Homosexualitat des Mannes und des Weibes. (Berlin, 1914) pp. 125-47.
- G. Henry, Sex Variants: a study of homosexual patterns. (Paul B. Hoeber, New York, 1941).
- C. H. Danforth, M. Trotter, Am J Phys Anthropol 5, 259 (1922).
- Q. Rahman, G. D. Wilson, Personal Individ Diff 34, 1337 (June, 2003)
Section 5.3 (Body-build)
6% of humans have a single scalp hair whorl rotation; about 92% have a clockwise scalp hair whorl rotation and the rest have a counter-clockwise scalp hair whorl rotation. Hair whorl rotation is prenatal in origin and does not result from combing hair. See Fig 1.
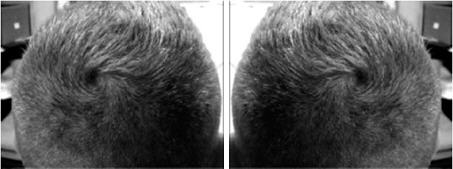
Fig 1: Clockwise scalp hair whorl rotation (left) and counter-clockwise scalp hair whorl rotation (right).(1)
Klar reported that in three samples of men containing an excess of male homosexuals, 29.8% (n = 272) had a counter-clockwise scalp hair whorl rotation compared to 8.2% of unselected male controls (n = 207) from the general population (p < 0.0001).(1) The samples enriched in homosexual men were obtained by observing men at Reheboth Beach, Delaware, which is a gay hangout. Klar has previously demonstrated an approximately 50% prevalence of counter-clockwise scalp hair whorl rotation among left-handers, as addressed in the book. Klar favors a genetic explanation of the association between left-handedness and counter-clockwise scalp hair whorl rotation as well as the association between male homosexuality and counter-clockwise scalp hair whorl rotation. Klar cites an old twin study by Bailey et al. which reported a roughly 50% concordance for homosexuality among male identical twins, but as discussed in the book, this study suffered from recruitment bias and newer studies on more representative samples of twins show a notably lower twin concordance for homosexuality that is either not greater than the concordance between two randomly chosen sex-matched individuals, as low as 13% or 20% for more exclusive homosexuality, and as high as 37% for any concordance for same-sex attraction.
Klar’s data constitute yet another piece of evidence of elevated phenotypic variability among male homosexuals and elevated atypical asymmetry among male homosexuals, and is consistent with the increased prevalence of non-right-handedness among male homosexuals, the association between left-handedness and developmental instability, and the association between homosexuality and developmental instability.
References:
- A. J. Klar, J Genet 83, 251 (Dec, 2004).
Section 5.4: Brain Structure
Kinnunen et al. compared alteration in brain glucose metabolism as a result of the administration of 40 mg of fluoxetine to 8 exclusively homosexual men and 7 exclusively heterosexual men (all except one heterosexual being consistently right-handed) in a double-blind placebo-controlled study.(1)
The researchers observed between-groups differential alteration in cerebral glucose metabolism (CMRglu) in several parts of the brain, including the hypothalamus. The between-groups difference in hypothalamic CMRglu is consistent with two previous reports of a smaller INAH-3 in male homosexuals compared to male heterosexuals and other differences reported in the literature such as a possible difference with respect to the suprachiasmatic nucleus, but the data obtained from Fig 2 below are unclear as to which regions of the hypothalamus are implicated.
Average CMRglu did not differ between treatments or groups. The alteration in CMRglu was more widespread among the homosexuals, as shown in Fig 1, which, according to the researchers, may represent greater heterogeneity among the heterosexuals (an apparently strange explanation, but then I am not familiar with PET scans), and an alternative possibility is that the homosexuals were less able to maintain stable brain functioning compared to the heterosexuals when challenged in a similar way (with respect to spatial distribution of altered brain function, not average CMRglu).
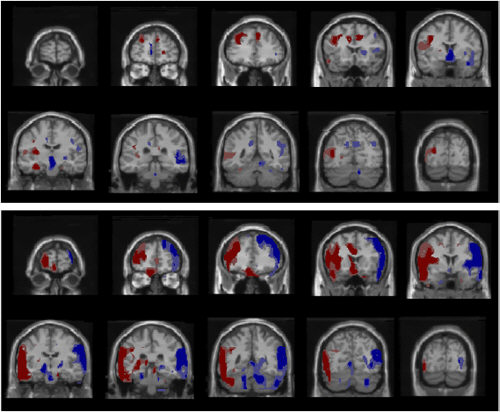
Fig 1: "Within-group activations. Heterosexual group shown in top panel, homosexual group in bottom panel. Increases in CMRglu in response to fluoxetine (relative to placebo) shown in red, decreases in blue. Darker colors significant at p less than or equal to 0.01, lighter colors at p less than or equal to 0.05. All images in neurological orientation."(1)
Fig 2 depicts the differential alteration in CMRglu between the groups.
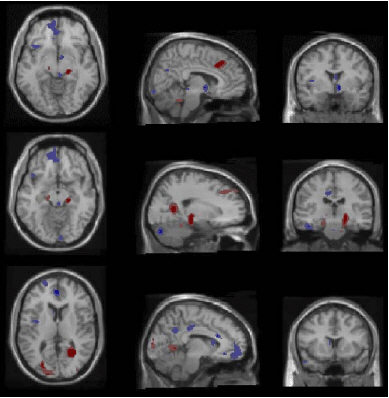
Fig 2: "Between-groups differential activation. Areas in which the heterosexual group, relative to the homosexual group, exhibited a significantly greater increase (or significantly smaller decrease) in CMRglu shown in red. Areas in which the homosexual group, relative to the heterosexual group, exhibited a significantly greater increase (or significantly smaller decrease) in CMRglu shown in blue. Darker colors significant at p less than or equal to 0.01, lighter colors at p less than or equal to 0.05. All images in neurological orientation." The homosexuals exhibited a smaller reduction in hypothalamic CMRglu compared to the heterosexuals. "The homosexual group exhibited significant increases in a portion of the prefrontal association cortex in which the heterosexual group exhibited no change, and in portions of the cingulate cortex in which the heterosexual group exhibited decreases. In contrast, the heterosexual group showed a significantly larger relative increase in lateral anterior cingulate, bilateral hippocampus/parahippocampal gyrus, and cuneate gyrus."(1)
Fluoxetine is an anti-depressant that belongs to the category of selective serotonin reuptake inhibitors (SSRIs). The book addresses evidence concerning increased serotonergic abnormalities among homosexuals and also mentions several studies addressing the favorable influence of SSRIs on several cases of paraphilias or sexual compulsion disorders and even the remission of homosexuality in rare cases.
References:
- L. H. Kinnunen, H. Moltz, J. Metz, M. Cooper, Brain Res 1024, 251 (Oct 22, 2004).
Section 5.7 (Homosexuality and Developmental Instability)
A simplified model of the relationship between homosexuality and increased odds of mental illnesses, as discussed in the book, should have been posted in the book, but here it is.
Section 5.7 (Homosexuality and Developmental Instability)
Qazi Rahman has again reported no differences between homosexuals and heterosexuals—both men and women—with respect to fluctuating asymmetry (FA).(1) This time he employed a better measure of FA, a composite measure based on width of ears, wrists, ankles and feet, and length of the ears and of the four fingers (excluding the thumb). However, he compared only 30 homosexual men, 30 heterosexual men, 30 homosexual women, and 30 heterosexual women; homosexuals being defined as those who scored 5-6 on the Kinsey scale and heterosexuals being defined as those who scored 0-1 on the Kinsey scale; the participants also self-identified as either homosexual or heterosexual. Given that FA is a poor indicator of developmental stability, Rahman did not have sufficient sample size to address possible group-level differences on this count. Besides, the homosexuals and heterosexuals in his sample did not differ with respect to handedness, body weight, or height, whereas the weight of prior evidence relates homosexuality to increased non-right-handedness (associated with developmental instability), male homosexuality to reduced body weight (less muscle mass, which is associated with disturbed development), and female homosexuality to increased body weight (excess body fat, which in turn is related to disturbed development). Rahman also made sure that none of the participants had a history of psychiatric/neurological illness, psychoactive medication/drug use, or injuries to hands or other features that would be measured for FA calculations. Once again, compared to heterosexuality, homosexuality has robustly been associated with a notably higher incidence of psychiatric illnesses and substance use, which is largely unaccounted for in terms of stigma, prejudice, and discrimination. Since some of the association between homosexuality and developmental instability would be accounted for by the association of homosexuality with elevated non-right-handedness, less muscle mass in men, excess body fat in women, higher incidence of psychiatric illnesses, and higher rates of substance use, Rahman’s sample, apart from being non-representative, is also deliberately less representative by design and hence more inadequate with respect to sample size than 30-member representative groups would be with respect to addressing developmental stability differences using the poor indicator of FA, i.e., this study does not even come close to undermining an abundance of evidence relating homosexuality to prenatal developmental disturbances.
On the other hand, Rahman found more masculine index finger-length to ring finger-length ratios (2D:4D) among both male and female homosexuals compared to their heterosexual counterparts, as has been documented previously, and also reported that a greater proportion of male homosexuals had older brothers. Rahman did not find any relation between 2D:4D and number of older brothers.
References:
- Q. Rahman, Psychoneuroendocrinology 30, 382 (May, 2005).
Section 5.7 (Homosexuality and Developmental Instability)
Consider additional references concerning comorbidity of psychiatric disorders; these references focus on comorbidity that is associated with bipolar disorder II (BP-II) [see: H. S. Akiskal, J Affect Disord84, 279 (Feb, 2005)], and are referenced because of the association between homosexuality and bipolarity:
Polydrug use is associated with bipolarity.[1] Depressed individuals with at least three anxiety disorder diagnoses (panic-agoraphobic, social phobic, and obsessive-compulsive (which is associated with homosexuality)) are commonly BP-II.[2] “Those with the triad of bulimic, atypical, and seasonal depressions also seem to belong to the realm of BP-II.”[3] “Related to the latter are depressions with rapid onset, short duration, and rapid offset, almost always associated with BP-II.”[4]
There is an association between bipolarity and anomalies such as borderline personality; compulsive disorders, including sexual compulsion; risky behaviors; and anti-social behaviors such as shoplifting, criminal sexual behaviors, and participation in riots.[5] The association between nonheterosexuality and borderline personality, risky behaviors, and sexual compulsion is well-documented.
“As far as erotic life [is concerned], depressed individuals who give history of having engaged, with more than occasional frequency, in sexual behavior with the same sex and both sexes at the same time or different times, are in our clinical experience, over-represented among the BP-II. Brief homosexual liaison during hypomania—sometimes protracted beyond—underscores the disinhibition that BP-II brings to the lives of these individuals (Akiskal and Pinto, 1999); alcohol use is often contributory to such behaviors.”[6]
References:
- {Akiskal et al., 1977 H.S. Akiskal, A.H. Djenderedjian, R.H. Rosenthal and M.K Khani, Cyclothymic disorder: validating criteria for inclusion in the bipolar affective group, Am. J. Psychiatry 134 (1977), pp. 1227–1233.}, {Angst et al., 2005 J. Angst, A. Gamma, V. Ajdacic, D. Eich, L. Pezawas and W. Rossler, Recurrent brief depression as an indicator of severe mood disorders. In: A. Marneros and F. Goodwin, Editors, Mixed States, Rapid Cycling and ‘Atypical’ Bipolar Disorder, Cambridge University Press, Cambridge (2005) In press.}, {Winokur, et al.,1998 G. Winokur, C. Turvey, H.S. Akiskal, W. Coryell, D. Solomon, A. Leon, T. Mueller, J. Endicott, J. Maser and M. Keller, Alcoholism and drug abuse in three groups-bipolar I, unipolars and their acquaintances, J. Affect. Disord. 50 (1998), pp. 81–89.}, {Akiskal and Pinto, 1999 H.S. Akiskal and O. Pinto, The evolving bipolar spectrum: prototypes I, II, III, IV, Psychiatr. Clin. North Am. 22 (1999), pp. 517–534.}, {Brady and Sonne, 1995 K.T. Brady and S.C. Sonne, The relationship between substance abuse and bipolar disorder, J. Clin. Psychiatry 56 (1995) (3 Suppl), pp. 19–24.}
- G. Perugi, H.S. Akiskal, S. Ramacciotti, S. Nassini, C. Toni, A. Milanfranchi and L. Musetti, Depressive comorbidity of panic, social phobic and obsessive-compulsive disorders: is there a bipolar II connection?, J. Psychiatr. Res. 33 (1999), pp. 53–61.
- G. Perugi and H.S. Akiskal, The soft bipolar spectrum redefined: Focus on the cyclothymic, anxious-sensitive, impulse-dyscontrol and binge-eating connection in bipolar II and related conditions, Psychiatr. Clin. N. Am. 25 (2002), pp. 713–737.
- {M. Strober and G. Carlson, Bipolar illness in adolescents with major depression: clinical, genetic, and psychopharmacologic predictors in a three- to four-year prospective follow-up investigation, Arch. Gen. Psychiatry 39 (1982), pp. 549–555.}, {H.S. Akiskal, P.W. Walker, V.R. Puzantian, D. King, T.L. Rosenthal and M. Dranonm, Bipolar outcome in the course of depressive illness: phenomenologic, familial, and pharmacologic predictors, J. Affect. Disord. 5 (1983), pp. 112–115.}
- {H.S. Akiskal, J.D. Maser, P. Zeller, J. Endicott, W. Coryell, M. Keller, M. Warshaw, P. Clayton and F.K. Goodwin, Switching from “unipolar” to bipolar II: an 11-year prospective study of clinical and temperamental predictors in 559 patients, Arch. Gen. Psychiatry 52 (1995), pp. 114–123.}, {S.L. McElroy, H.G. Pope Jr., P.E. Keck Jr., J.I. Hudson, K.A. Phillips and S.M. Strakowski, Are impulse-control disorders related to bipolar disorder?, Compr. Psychiatry 37 (1996), pp. 229–240.}, {G.B. Cassano, S.L. McElroy, K. Brady, W.A. Nolen and G.F. Placidi, Current issues in the identification and management of bipolar spectrum disorders in ‘special populations’, J. Affect. Disord. 59 (2000), pp. S69–S79.}, {J. Deltito, L. Martin, J. Riefkohl, B. Austria, A. Kissilenko, C. Corless and P. Morse, Do patients with borderline personality disorder belong to the bipolar spectrum?, J. Affect. Disord. 6 (2001), pp. 221–228.}
- H.S. Akiskal and O. Pinto, The evolving bipolar spectrum: prototypes I, II, III, IV, Psychiatr. Clin. North Am. 22 (1999), pp. 517–534
Section 5.8 (Bisexuality)
Here are the details of a study examining retrospective assessments of changes in the components of sexual orientation: behavior, fantasy, attraction, and identity.(1)
The participants were a non-random sample of 420 men and 342 women, aged 36-40 years, recruited to answer a sexuality/sexual-orientation-across-the-lifespan survey via advertisements in print publications, announcement to community groups, and the internet. Of the 513 packets sent to 15 community groups for distribution, 26.7% were returned, and of the 73 phone requests for questionnaires to be sent by mail, 32.9% were returned.
Table I shows the distribution of those that self-identified as homosexual, heterosexual, or bisexual.
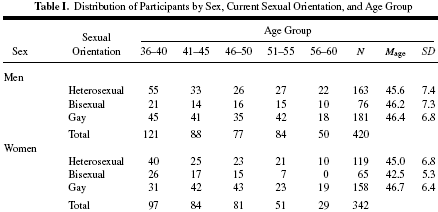
Here is how the authors derived change scores; the Kinsey scale is a 7-point scale ranging from 0-6:
Here is how the authors derived the total change in lifespan (TCL):
Since the opportunity for change was not equal across the ages, age was treated as an independent variable.
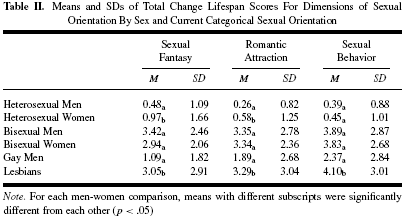
Table II shows that the TCL was greater among the self-identified nonheterosexuals.
No change ever in any component was reported by 66% of self-identified heterosexual men, 51% of self-identified heterosexual women, 33% of self-identified homosexual men, 9% of self-identified homosexual women, 5% of self-identified bisexual men, and 1.5% of self-identified bisexual women. The prevalence of lifetime-exclusive heterosexuals—based on random, population-based samples—is much greater (about 80%) than what these figures suggest, but then a sexuality survey disproportionately draws responses from the sexually atypical and the study had a low response rate plus was non-random to boot.
At most a one-point cumulative shift in lifetime within any component was reported by 93% of self-identified heterosexual men, 86% of self-identified heterosexual women, 48% of self-identified homosexual men, 20% of self-identified homosexual women, 10% of self-identified bisexual men, and 9% of self-identified bisexual women.
Table III shows the shifts in sexual orientation identity over lifetime.

These data are in agreement with a considerable amount of information that I have documented in my book: heterosexuality is a more developmentally stable outcome than nonheterosexuality; sexual orientation taxa (categories) with respect to heterosexual and homosexual interests are dichotomously classified as heterosexual vs. nonheterosexual; nonheterosexuals are more mentally unstable than heterosexuals, bisexuals are more mentally unstable than homosexuals, and women are more mentally unstable than men; the exclusively homosexual are less prevalent among women than men; and multiple erotic target shifts characterize very few people, thereby arguing against the flexibility, malleability, fluidity, or plasticity of sexual orientation since such labeling would require one to demonstrate back and forth shifts in many individuals. The authors refer to the changes examined as flexibility of sexual orientation; others may use malleability, fluidity, or plasticity, instead of flexibility, as previously mentioned, but the phenomena described are best addressed as one of mental instability that is eventually resolved in several individuals.
The lack of sex differences among self-identified bisexuals compared to self-identified homosexuals or self-identified heterosexuals has prompted the authors to propose that bisexuals may be qualitatively distinct from homosexuals and heterosexuals. However, taxometric analyses and various considerations suggest that bisexuals and homosexuals, although different, belong to the same taxon (category).
References:
- K. K. Kinnish, D. S. Strassberg, C. W. Turner, Arch Sex Behav 34, 173 (Apr, 2005).
Section 5.9 (Homosexuality and Twinning)
The book discusses several examples of discordance for physical traits within pairs of identical twins. See two examples of major right-left discordance for physical traits in the same individual below.(1)
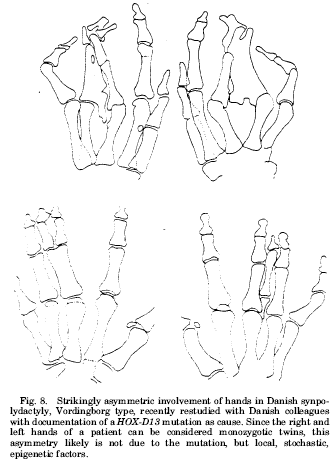
References:
- J. M. Opitz, A. Utkus, Am J Med Genet 101, 359 (2001).
Section 5.10 (Is Homosexuality a Mental Illness?)
This section features the following passage:
Additional evidence concerning the above:
References:
- H.S. Akiskal and K. Akiskal, Re-assessing the prevalence of bipolar disorders: clinical significance and artistic creativity, Psychiatr. Psychobiol. 3 (1988), pp. 29s–36s.
- H.S. Akiskal and K. Akiskal, Cyclothymic, hyperthymic and depressive temperaments as subaffective variants of mood disorders. In: A. Tasman and M.B. Riba, Editors, Annual Review vol. 11, American Psychiatric Press, Washington, DC (1992), pp. 43–62.
- R. Richards, D.K. Kinney, I. Lunde, M. Benet and A.P. Merzel, Creativity in manic-depressives, cyclothymes, their normal relatives, and control subjects, J. Abnorm. Psychology 97 (1988), pp. 281–288.
- N.C. Andreasen, Creativity and mental illness: prevalence rates in writers and their first-degree relatives, Am. J. Psychiatry 144 (1987), pp. 1288–1292.
- K.R. Jamison, Manic-depressive illness and creativity, Sci. Am. 272 (1995), pp. 62–67.
Section 5.16 (The Genetics of Homosexuality and Other Unusual Sexual Interests)
Two studies addressing the number of offspring among the relatives of male homosexuals are addressed in this update. These studies address the question whether the genetic factors associated with male homosexuality increase the number of offspring among the heterosexual relatives of male homosexuals, thereby explaining how genes for male homosexuality could persist.
The first study comes from Northern Italy. The authors compared 98 homosexual men and 100 heterosexual men who were quizzed on demographics and sexual orientation of their siblings, first cousins, parents, uncles, and grandparents (a total of about 4,600 individuals).(1) The authors reported that compared to heterosexual men, male homosexuals reported familial clustering of male homosexuality among their maternal relatives (Table 1), something that has been reported previously, and also tended to be later born, especially with respect to sons, which is a robust correlate of male homosexuality; the homosexual men also had an excess of brothers among their older siblings. 21% of the variance in sexual orientation was accounted for by the “number of male homosexuals among maternal relatives” (14% of the variance) and “number of older brothers” (6.7% of the variance).(1)

Note from authors concerning Table 1: “ In table 1, it should be noted that the higher total number ( N ) in the paternal line than in the maternal line naturally emerges from the following. (i) The presence of one more kin class in the paternal line (sons of paternal uncles); while in the maternal line the corresponding kin class (sons of maternal uncles) could not be included because the members of this kin class do not share the X chromosome with our subjects. (ii) The fact that the number of fathers classified in the paternal line is higher than the number of brothers classified in the maternal line, as expected in a population with low general fecundity, such as the studied population from northern Italy. We also included fathers in the analysis, as they could, in principle, be homosexual; homosexuals are reproductive and can transmit genetic factors (Moran 1972; Bell & Weinberg 1978).”(1)
The authors found that the maternal relatives but not paternal relatives of homosexual men, especially mothers and maternal aunts, had more children than the maternal relatives of heterosexual men (Table 2).
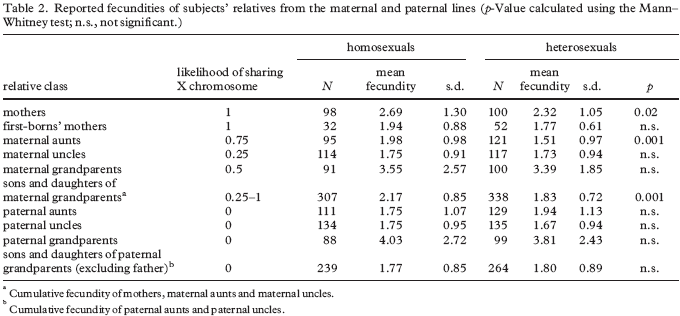
Since male homosexuals tend to be later born, especially with respect to sons, women with a greater number of offspring will be producing a relatively greater proportion of male homosexuals, and this need not imply that some of the genes that increase the odds of a male homosexual outcome also increase female fertility. Similarly, as addressed in the book, there is a tendency for homosexuality to cluster into some families, which appears to be related to genetic rather than familial environmental factors. Therefore, a combination of these genetics and another-factor-related tendency for women in some families to have more children could combine to explain the data in this study, i.e., one does not have to conclude that the genetics behind male homosexuality also increase female fertility. On the other hand, the authors reported a non-significant trend toward a greater number of children on the part of mothers whose first-born child happened to be a male homosexual. It is not clear whether this trend would have been significant had the sample size been larger, but assuming that it would have been significant and that this correlate happens to be genuine, then the results could possibly be accounted for via higher levels of estradiol among the mothers/aunts of male homosexuals since estradiol levels are strongly related to female fertility and because higher prenatal testosterone exposure is implicated in male homosexuality, which should also correspond to higher testosterone-derived-estradiol exposure, i.e., male homosexual outcomes could be related to the disruptive effects of excess estradiol exposure, which defeminizes/masculinizes a variety of mammalian behaviors during early development, likely also in humans as addressed in the book. Therefore, once again, the results cannot be used to claim evidence for genetic factors behind male homosexuality that also increase female fertility.
An alternative exotic possibility that could account for a relation between the fertility of maternal relatives and male homosexuality is developmental interference caused by sexually antagonistic genes in mitochondrial DNA (inherited from the mother).(2) As mentioned in the book, sexually antagonistic genes benefit one sex but harm the other sex, and if such genes are implicated, then the relation is certainly not accounted for within a developmental-stability framework. On the other hand, since Table 2 shows increased fertility among mothers and maternal aunts but not decreased fertility among the maternal uncles of male homosexuals, the relevant mitochondrial DNA variant would be expected to be quite common, and would not by itself be leading to male homosexuality since male homosexual outcomes are uncommon.
The data in this study are inconsistent with several hypotheses on the genetic bases of male homosexuality.(1) For instance, if a single-locus genetic model [nuclear genes] is assumed that explains male homosexuality as a result of homozygosity, then the differences between the maternal and paternal pedigree lines and the clustering of male homosexuality among maternal relatives could not be explained.(1) If the single-locus genetic model postulates an X-linked allele, then since the data (Table 2) do not show reduced number of offspring among the maternal uncles of male homosexuals, five of whom were childless homosexuals or bisexuals, but show enhanced fertility among mothers and maternal aunts instead, then this allele would be common, but male homosexuality is uncommon and the families of heterosexuals should also be expected to have a high incidence of this allele, which would not explain the between-groups difference in fertility of maternal relatives.(1) A polygenic inheritance of male homosexuality as proposed by Miller(3) would also not explain the differences between the maternal and paternal pedigree lines and the clustering of male homosexuality among the maternal relatives of male homosexuals.(1) Miller’s proposal may account for the data if some of the alleles are X-linked, but as addressed in the book, Miller proposes male homosexual outcomes to result from an excess of feminizing alleles, which is inconsistent with numerous hypermasculine correlates of male homosexuality in addition to the normomasculine, feminine, and hypomasculine correlates, a paradoxical mix that can be parsimoniously explained in terms of male homosexuality resulting from developmental disturbances caused by excess prenatal exposure to masculinizing factors, the genetic bases for which should not favor female fertility.
The second study comes from London. This study assessed—via anonymous questionnaires—301 homosexual men and 401 heterosexual men who attended two central London STD clinics.(4) The men were drawn from consecutive male clinic attendees. A total of 1,061 men answered the questionnaires but non-white men were excluded because their family size was a lot larger than that of white men and 72.8% of non-white men described themselves as heterosexual whereas 55.3% of white men described themselves as heterosexual.
The homosexual men were older by about 5 years, on average, more likely to be Protestant, less likely to be employed, and more likely to be involved in non-manual occupations. Table II shows comparisons of the family size data between the two groups as it was and after adjustment for sociodemographic factors.
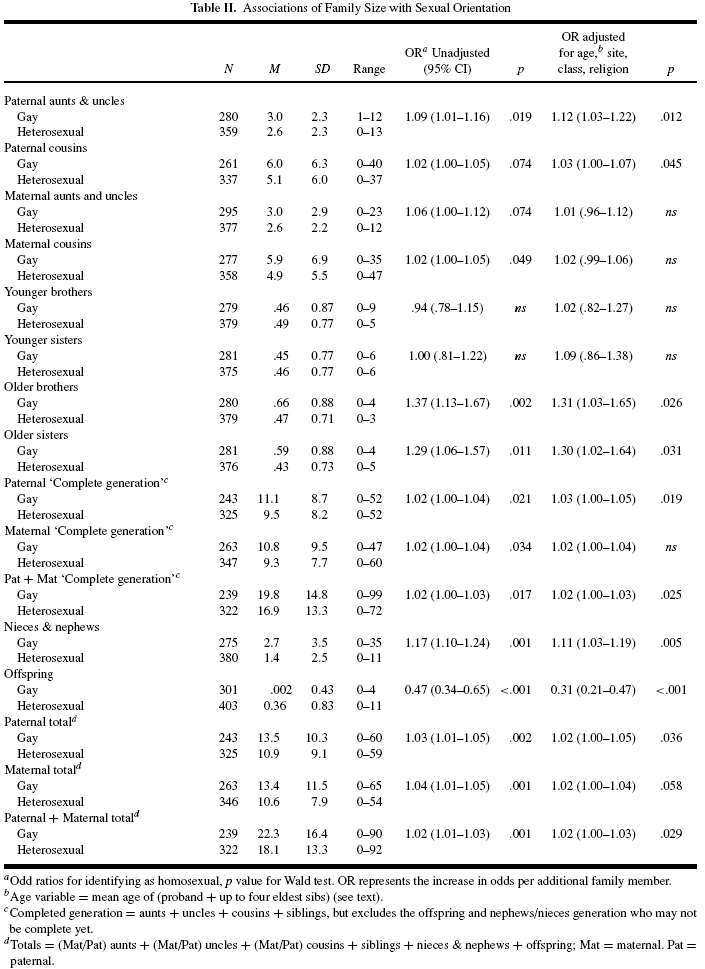
Note from authors concerning superscript “b” in Table II. “To adjust for overall “family age,” we used a form of mean sibling age, taking the mean age of the participant and up to his eldest four siblings. This aggregate age measure was a more powerful confounder of associations between family size and sexual orientation. A limit of the four eldest siblings was set to minimize the number of participants excluded due to missing data.”(4)
As seen in Table II, the homosexual men had a greater number of both older brothers and older sisters and came from larger families. Once again, it cannot be assumed from such data that some genetic factors increasing the odds of male homosexual outcomes also increase fertility among the heterosexual relatives of homosexual men since homosexual men, tending to be later born, will disproportionately come from families having larger sizes.
References:
- A. Camperio-Ciani, F. Corna, C. Capiluppi, Proc R Soc Lond B Biol Sci 271, 2217 (Nov 7, 2004).
- J. A. Zeh, D. W. Zeh, Trends Genet 21, 281 (May, 2005).
- E. M. Miller, Arch Sex Behav 29, 1 (2000).
- M. King et al., Arch Sex Behav 34, 117 (Feb, 2005).